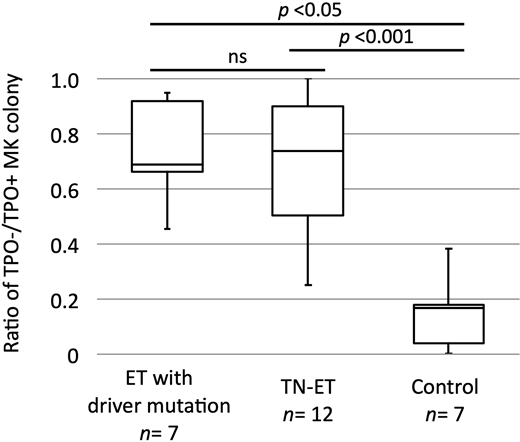
Somatic mutations in JAK2, MPL, and CALR are found in approximately 80% of patients with essential thrombocythemia (ET), whereas the remaining patients are negative for disease-defining mutations and are defined as triple-negative (TN). Studies have shown that some patients with TN-ET harbor non-canonical mutations in JAK2 and MPL; however, the failure to identify recurrent mutations in most patients has made the pathogenesis of TN-ET ambiguous (Milosevic Feenstra et al. Blood 2016, Cabagnols et al. Blood 2016). In this study, we screened 483 patients suspected as having ET in a single center, performed mutation analysis for JAK2 V617F, CALR exon 9, and MPL exon 10, and centrally reviewed bone marrow specimens. We identified 23 patients with TN-ET based on the WHO 2016 criteria. Sequencing analysis of these patients revealed non-canonical mutations in JAK2 and MPL in 4 cases. Whole exome-sequencing analysis of genomic DNA from peripheral blood and CD3-positive cells from 9 patients revealed that 2 patients harbored somatic mutations in other genes; 7 patients showed no detectable somatic mutation. A STAT5 reporter assay revealed that unlike JAK2 V617F and MPL W515L, all non-canonical mutants of JAK2 or MPL activated STAT5 similar to wild-type proteins, suggesting that these mutations did not drive the disease. Statistical analysis of clinical records revealed that patients with TN-ET were mostly young (median age of 36.0 years), female (18/23, 78.3%), and had neither a history of thrombosis nor progression to secondary myelofibrosis and leukemia, demonstrating the unique characteristics of TN-ET. The presence of clonal hematopoiesis, analyzed using genomic DNA purified from granulocytes of peripheral blood from female patients in a human androgen receptor assay, revealed that only 1 out of 15 patients was clonal. Hypothesizing that TN-ET was reactive thrombocytosis, the concentrations of cytokines promoting platelet production such as thrombopoietin (TPO) and interleukin-6 (IL-6) in the serum were analyzed. However, no significant differences in concentrations were observed among ET with driver mutations, TN-ET, and healthy individuals. We next examined the capacity of hematopoietic stem cells from patients with TN-ET to form megakaryocytic colonies. CD34-positive cells purified from cryopreserved bone marrow cells were cultured in the absence or presence of TPO. CD34-positive cells derived from patients with TN-ET exhibited an equivalent capacity to form megakaryocytic colonies compared to those from patients with ET harboring a driver mutation, even in the absence of TPO (Figure 1). Thus, in TN-ET, megakaryopoiesis may have been induced in a cell-autonomous manner. In 10 patients with TN-ET with available blood count data, no sign of thrombocytosis was observed before ET development, indicating that thrombocytosis was not hereditary but rather occurred via an alternate mechanism, such as aberrations in epigenomic regulation that induced cellular transformation (Ohnishi et al, Cell 2015). Taken together, TN-ET is a distinctive disease entity associated with polyclonal hematopoiesis and paradoxically caused by hematopoietic stem cells harboring a capacity for cell-autonomous megakaryopoiesis.
Komatsu:Takeda Pharmaceutical Co., Ltd, Novartis Pharma KK, Shire Japan KK: Speakers Bureau; PPMX: Consultancy, Research Funding; Meiji Seika Pharma Co., Ltd.: Patents & Royalties: PCT/JP2020/008434, Research Funding; AbbVie: Other: member of safety assessment committee in M13-834 clinical trial.; Otsuka Pharmaceutical Co., Ltd., Shire Japan KK, Novartis Pharma KK, PharmaEssentia Japan KK, Fuso Pharmaceutical Industries, Ltd., Fujifilm Wako Pure Chemical Corporation, Chugai Pharmaceutical Co., Ltd., Kyowa Hakko Kirin Co., Ltd., Takeda Pharmaceutica: Research Funding; Otsuka Pharmaceutical Co., Ltd., PharmaEssentia Japan KK, AbbVie GK, Celgene KK, Novartis Pharma KK, Shire Japan KK, Japan Tobacco Inc: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal